Break the chain of hospital visits. CAR-T is changing myeloma treatment

Published April 27, 2026 07:18
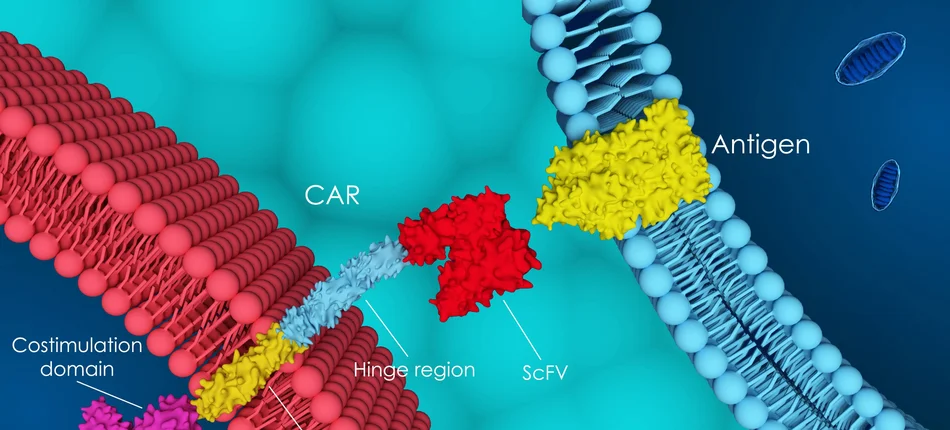
The groundbreaking CAR-T therapy is reimbursed in Poland for patients with acute lymphoblastic leukemia and lymphomas. The therapy has also been registered for the treatment of plasmocytic myeloma. It works so effectively that, according to doctors, some myeloma patients can be completely cured.
Own lymphocytes to produce the drug
CAR-T therapy involves the patient receiving his or her own T lymphocytes - immune system cells - that have been appropriately genetically modified in the laboratory. It is therefore a "living medicine". - These are cells present in everyone's body. They are responsible for anti-tumor and anti-infective immunity. By performing allogeneic transplants, we have seen that T cells are crucial to a patient's recovery from cancer. Their genetic modification makes them more aggressive against cancer cells. They are able to recognize them, bind to them and destroy them," explains Prof. Lidia Gil of the Department of Hematology, Transplantation and Cell Therapy at the Poznan University of Medical Sciences. She adds that lymphocytes after genetic modification have the ability to multiply. - The drug does not wear off completely, as it has the ability to expand, she explains. The expert reports that it takes about two to three hours to collect lymphocytes from a patient. Then they are packaged, picked up by a courier and sent to a specialized laboratory. Genetic modification for myeloma takes about four weeks. - The material arrives frozen and is already a drug that we administer to the patient," he says.
Since registration in Europe in 2018, more than 17,000 such procedures have been performed.
How was Emili cured after 6 years of ineffective therapies?
The breakthrough for CAR-T therapy came in 2012, when 12-year-old Emily Whitehead of Pennsylvania, who at age 5 developed lymphoblastic leukemia resistant to all forms of treatment, was given modified lymphocytes of her own as part of a clinical trial. The team of pediatric oncologist Dr. Stephen Grupp, director of the Children's Cancer Research Center in Philadelphia, administered the cells to her without preceding it with any other therapy and without adding any other therapy after. The girl achieved remission of the disease and is still healthy today. She never received anticancer treatment after that. She created a foundation that today supports the development of innovative methods in oncology. In Poland, the first lymphoma patient received CAR-T in 2019, and two years later it was administered for the first time in Poland to a patient with plasmocytic myeloma. The patient received it as part of a clinical trial.
Flight to Mars
- When I first heard about this therapy, I thought we were talking about a trip to Mars. And I absolutely did not even dream that it would be available in our country," recalls Prof. Lidia Gil. - I met with the manufacturers of this drug on the occasion of various foreign conferences. I asked them not "if" but "when" they planned to expand the use of this drug to Poland," she says. - I finally managed to arrange a working lunch with them and we agreed that they would come to Poland to see what potential we had. After returning to Poznan, I gathered my transplant team and we decided to prepare, Half of us didn't believe that the Americans would come. But they came and so it began. It was January 2019 and from that moment the whole accreditation pathway was planned for us," says the hematologist. - Today we are the largest center in Poland and not only, because in Switzerland or Austria, for example, they have less experience and fewer procedures performed. And we have already cured a great many patients. Today we apply this treatment to very well selected patients. We know who to qualify for the treatment, how to administer it, how to manage side effects. We were frightened by them, but they turned out to be less dangerous than those we see with transplantation," he points out. - Although we have learned a lot, we still feel that we are at the beginning of the road, as the technology is developing strongly, he adds.
Only 3-year survival
Plasmocytic myeloma is a cancer of plasma cells-an element of the immune system that are responsible for, among other things, producing antibodies. In myeloma patients, they proliferate uncontrollably, cease to play their role and produce abnormal proteins that can damage other organs and tissues, such as kidneys and bones. In Poland, myeloma is detected in about 2,000 people each year.
More than 20 years ago, the median survival time for these patients was three years. - Today the median survival is 10-15 years, but there are patients, and we have some in our clinic, who are living with the disease for more than 20 years," says Prof. L.Gil.
Nowadays, plasmocytic myeloma has become a chronic disease. Patients are living longer, but they are constantly in need of specialist consultations and successive lines of therapy. This often makes them simply tired of treatment. - In addition, at a certain stage the options for effective treatment are exhausted, patients lose response to this treatment," says Prof. Gil.
Prof. Lidia Gil recalls her first patient with plasmacytoid myeloma. - This was a patient with very severe bone pain, weak, with recurrent infections, literally a hunched over man in pain. Applying the first line of treatment caused all these ailments to regress in a very short time. I was extremely proud that as a young doctor I was successfully treating a cancer patient. Only that this response lasted several months, and then I experienced the passing of this man together with his family. The median survival at the time was 3 years, but it was really much shorter. Today, I have patients I started treating 20 years ago, and this prolongation of survival is indeed spectacular," he says. However, these patients still require further therapies. So we need a treatment that will break this chain that causes the patient to be constantly linked to the hospital," he urges.
Effectiveness of CAR-T
The CAR-T method was initially used for patients who were past many treatments and had exhausted other therapeutic options. - Even in this group, we obtain long-term periods of improvement. However, we will undoubtedly get even better results when CAR-T is administered to patients at earlier stages of treatment. There are patients in whom this therapy gives very good results when given in the second line of treatment," says Prof. Lidia Gil.
CAR-T therapy should be given to myeloma patients who experience relapse, resistance to first-line therapy. - Perhaps in the future we will use this method for patients with residual disease, that is, patients in whose body after treatment a very small number of tumor cells are present, detected by very sensitive methods. In these patients, sooner or later - more likely sooner - there will be a relapse. It seems that CAR-T therapy for these patients would be an ideal solution," says Prof. Gil.
Studies are currently underway that will answer the question of whether CAR-T treatment will replace hematopoietic cell transplantation and be part of first-line therapy. - I am sure that myeloma will be a curable disease, I just don't know when it will happen. I think that when it comes to the use of CAR-T therapy in plasmocytic myeloma, we are at the beginning of the road. We are just starting to administer these therapies at earlier and earlier stages of treatment, and not all the studies have been completed, but this technology will improve. In my opinion, there is no turning back from CAR-T, and I am convinced that malignancies in hematology will be curable with CAR -T therapies," he points out.
Accessibility for Polish patients
- We would like to see CAR-T therapies reimbursed. We realize that they are expensive, but experience in treating patients with lymphoma or acute lymphoblastic leukemia shows that their use pays off. CAR-T is administered to a patient once and is a one-time expense, while the other therapies are continuous therapies that we use on patients as long as patients respond to treatment or until side effects develop. On top of that, they require the continuous involvement of the team at the hematology centers, because the patients on whom we use them come every two or three weeks for a long time," says Prof. Gil.
Jakub Gierczynski, a health care expert, recalls that the AOTMiT has issued a positive opinion for reimbursement of this therapy for the treatment of relapsed and refractory plasmocytic myeloma from the second line of treatment.
- 30% of patients are under the age of 65, and this is also an argument. Another is the limited population of patients eligible for this therapy. Besides, the Polish Society of Hematology and Transfusion in TOP HEMATO has placed this therapy in the top ten of those that should be reimbursed. It is also recommended by national consultant in hematology Prof. Ewa Lech-Marańda . The NHF report done together with hematologists on plasmocytic myeloma and the analysis of the B54 drug program shows tools to control and monitor the expensive treatment. And in the report, "Needs and Expectations of Patients with Relapsed Plasmocytic Myeloma," surveyed patients, half of whom were between the ages of 50 and 59, said that the biggest traumatic patient experience is relapse, he pointed out. - CAR-T therapies potentially provide tangible savings and health and social benefits over the long term. A single administration can replace years of costly treatment with multiple lines of cancer therapies, reduce hospitalizations, and allow patients to return to full work and life activities. Despite the high cost of a single administration over a multi-year horizon, this therapy may be less of a burden on the system than repeatedly funding standard treatment regimens for the care of patients with advanced disease, he added.
We are prepared to treat
Poland is organizationally prepared to implement CAR-T therapy in plasmocytic myeloma. Currently, there are 15 centers in the country administering CAR-T therapy - 13 facilities treat adults, and two - in Wroclaw and Bydgoszcz - treat children. Three more centers - in Krakow, Torun and Gorzow - are waiting for accreditation, without which it is not possible to start treating patients with the cell-based method. To date, more than 650-700 patients have been treated in Poland with CAR-T therapy for other hematological indications, mainly lymphoma and acute lymphoblastic leukemia. This experience provides a solid basis for expanding the indication to include plasmocytic myeloma. Prof. Lidia Gil assures that the Ministry of Health and the Fund have very good control over patients who receive CAR-T therapy, thanks to the functioning of a coordination team with representatives from the Ministry and the Fund. In Poland, about 50 patients could benefit from CAR-T therapy for plasmocytic myeloma.
Topics
limfocyty T / refundacja terapii / prof. Lidia Gil / szpiczak plazmocytowy / CAR-T / refundacja / lidia gil / terapia komórkowa / Jakub Gierczyński